Aeterna Health's Stem Cell (svCell) Application is an emerging therapy that utilizes live cell clusters with the potential to promote healing and rejuvenation. By harnessing the regenerative properties of stem cells, svCell therapies are being studied for their ability to support tissue repair, reduce inflammation, and improve overall health, though individual results may vary.
Osteoarthritis is a degenerative joint disease causing the breakdown of cartilage and subchondral bone, leading to pain, swelling, and limited mobility. The disease also triggers inflammation that further damages the cartilage. As it progresses, bones can break down and form spurs, and in severe cases, the cartilage may wear away completely, causing bone-on-bone contact.
Commonly affected areas include the knee, hip, back, and small joints in the hands and feet, often due to repetitive movements or injuries. Symptoms include persistent joint pain, tenderness, stiffness, locking, loss of motion, and swelling.
svCell therapy is a cell-based treatment that utilizes adipose-derived stromal vascular fraction (SVF) to manage osteoarthritis. It may be offered under medical supervision in an accredited day hospital setting. svCell therapy is a complementary treatment option that is being researched for its potential to support the body’s natural healing processes and reduce inflammation, which may help alleviate osteoarthritis symptoms, improve mobility, and promote cartilage restoration.
The goal of svCell therapy for osteoarthritis is to provide an alternative option between traditional pain management and joint replacement surgery. However, its use and effectiveness are subject to ongoing research, and the therapy should only be administered by a qualified healthcare provider in compliance with regulatory guidelines.
svCell is an adipose-derived stromal vascular fraction containing a multipotent heterogeneous mix of regenerative cells that can help reduce inflammation, promote healing mechanisms, and improve tissue repair.
svCell therapy for osteoarthritis has the potential to promote self-healing through the following mechanisms:
svCell therapy aims to reduce joint pain and inflammation while maintaining – and in some cases, improving – joint function and reducing stiffness, which may postpone or avoid joint replacement surgery in the future.
Pain and Function
Pain and function improvement demonstrated with rapid post-treatment effect, enduring for 60 months.
Changes in the VAS score and the WOMAC score during the 5 years before and after treatment in the SVF group and control group.
A The mean VAS score change. B The mean WOMAC total score changes. *p value <0.05, **p value < 0.01,***p value < 0.001, ns, non-significant (p value > 0.05), compared with pre-treatment.
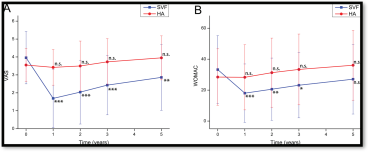
Image derived from Zhang et al. 2022
Inflammation
The marked decrease in catabolic and pro-inflammatory molecules (MMP-2, IL-1B, IL-6, IL-8) and a significant increase in anabolic and anti-inflammatory molecules (IGF-1 and IL-10).
Evolution of different markers in knee OA treatment and SVF cell characterization. B.L., Baseline; M, months; black
stars indicate significant differences (p < 0.05, non-parametric Wilcoxon test) Image derived from Lapuente 2020
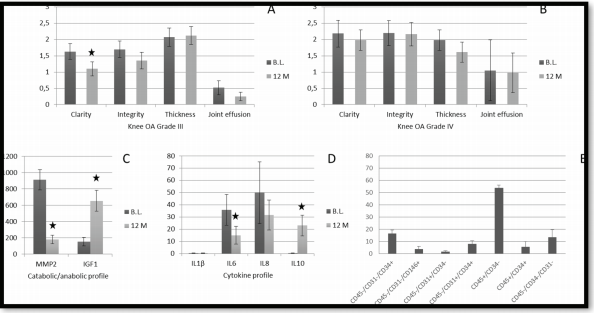
Image derived from Lapuente 2020
In some cases, osteoarthritis treatment may involve surgery (hip joint or knee replacement) to replace the joints. Cell therapy technology may provide an alternative approach for certain patients.
| Parameter | Knee Replacement | svCell Therapy |
| Impact on bones |
Inevitable loss of bone: Tibia, Femur & Patella |
Injection only - no bone loss |
| Mechanism | Prosthesis | Natural regenerative cells |
| Invasion | Highly invasive | Minimally invasive |
| Anesthesia | General/Spinal | Local |
| Recovery | Extended recovery | Easier & quicker |
| Treatment type | Artificial implant | Autologous cell |
| Nature of intervention | Mechanical | Biological |
| Indication |
1. Gr- IV OA 2. Not suitable forRA Patients |
3. All Gr. of OA** 4. Suitable for RA also |
| Revision surgery | Difficult with poor outcome | Easy |
| Need for Revision Surgery | Generally required after 10-15 years * | Need may not be there* |
| Effect on lifestyle | Numerous restrictions | Substantial improvement |
| Hospitalization | Minimum stay of 4-5 days | Out patient |
| Implant Rejection | Possibility | No rejection (as autologous cells used) |
| Effect on cartilage regeneration | It doesn’t treat the underlying cause (i.e. cartilage degeneration) | It treats underlying cause by aiding cartilage regeneration and immunomodulation of inflammation |
| Effect of body weight on outcome | Poor result in case of obese patients | Effective for all body weights |
| Age criteria | Usually avoided for younger patients | Suitable for all age groups |
| Fitness for surgery |
Patients must have good fitness to undergo this surgery |
It is not a major limiting factor |
svCell therapy, is an Australian patented biotechnology process, that has been used as a complementary approach to treat osteoarthritis in Australia and the United States for many years.
The potential benefits of using svCell therapy in the clinical treatment of osteoarthritis in Australia include:
as the svCell ultrasonic cavitation process uses a person’s own cells, there is a reduced risk of infection or disease transmission;
as the svCell concentration process does not culture cells, there is no risk of rejection or allergic reaction.
svCell therapy has the potential for therapeutic advantage as it contains a mix of regenerative cells, with some scientific research suggesting that this mixed cell population may provide superior treatment.
Autologous adipose-derived svCell therapy has a reduced risk of adverse reactions or serious side effects.
As the treatment is autologous, with the svCell regenerative cells derived from the patient’s own body, there is a lower risk of infection or disease transmission. Using the patient’s own, uncultured cells reduces potential risks and complications as associated with rejection and allergic reactions.
The ultrasonic cavitation concentration process does not use any enzymes or animal products.. Within a relatively short period of time, potentially within 4 hours, the patient’s cells are concentrated and injected. As a result, there is a reduced chance of infection, disease transmission, bacterial contamination, or allergic reaction.
Over the last 12 years, there has been a body of evidence in peer-reviewed scientific literature supporting the safety of svCell in the treatment of osteoarthritis for knee and hip disease.
The svCell therapy procedure involves removing adipose (fat) tissue from the patient’s abdomen, concentrating a mixture of cells, and then re-injecting the cell concentration into the patient’s painful joint(s).
In Australia svCell therapy procedures must be conducted within an accredited hospital setting under medical supervision with patient’s admitted to the hospital as a day surgery patient. The procedure is usually completed within 4 hours after which the patient may go home.
Preparation
Patients taking Aspirin, anti-inflammatory medications (such as Voltaren and Mobic), corticosteroids, or herbal medications (e.g., Krill oil, fish oil, Vitamin E, and Ginkho Biloba) are advised that these must be discontinued at least 10 days before the procedure, according to standard hospital admission instructions. Those patients taking aspirin (Cartia, etc.), clopidogrel (Plavix or Iscover), warfarin, or anticoagulants for a heart condition or stroke prevention, would be provided specific information by their own doctor on when to stop and restart these medications.
Removal of adipose tissue
Patients receive oral sedation, and local anesthetic, and may be prescribed an antibiotic to reduce the risk of infection and are expected to be comfortable, and relatively pain-free.
A syringe or miniliposuction method is used to extract 60-300ml of adipose (fat) tissue from the patient’s abdomen, as that is all that is required. The amount of fat consumed will be determined by the number of joints to be treated. The treating doctor will explain the method of fat extraction to be used prior to the procedure. Because the procedure is minimally invasive, no stitches are usually needed.
Collect regenerative cells
Following adipose tissue removal, a sterile laboratory technician immediately performs the svCell ultrasonic cavitation process to concentrate the patient’s regenerative cells. A flow cytometer is used to count cells and viability and determine the best dosage for the injection of svCell concentrate.
Injection
The injection site is prepared in advance. A Doctor will then inject the svCells into the joint(s).
Where necessary, ultrasound guidance can be used to guide needle placement.
After the injection, patients are monitored for 1 hour before being released to go home.
Please Note: The effectiveness of svCell/iCell therapy can vary based on individual circumstances. We encourage discussions with our medical team to understand how svCell/iCell therapy might align with your needs. Compliance with TGA regulations, Aeterna Health is committed to adhering to the Therapeutic Goods Administration (TGA) regulations. All potential benefits mentioned are based on ongoing research patient safety, and informed decision-making.
To learn more about the services offered and the work Aeterna Health is involved with, please complete the form below.
Suite 1, 990 Toorak Road, Camberwell VIC 3124
Grayclay College of Health
Level 1, 1 Short Street, Southport QLD 4215
+61 3 7016 0801
+61 7 5531 3738
info@aeternahealth.com.au
